 |
 |
AbstractPurposeDifferentiation of urinary tract infection (UTI) from viral infection is a critical challenge in febrile children in emergency departments (EDs). This study aimed to assess the predicting performances of creatinine, C-reactive protein (CRP), and white blood cell (WBC) for predicting UTI in the children.
MethodsThis study was a retrospective analysis of a prospectively enrolled cohort of febrile children who presented to our children’s hospital ED from August 2016 through February 2018. We included previously healthy, febrile (≥ 38。C) children younger than 24 months whose urine cultures were obtained. Accuracy of creatinine, CRP, and WBC were assessed by optimal cutoffs, which were calculated using receiver operating characteristic curves.
ResultsAmong the total 33,013 children to the ED, 7,847 (23.8%) febrile children were registered to the fever registry. Finally, 506 children were included, and UTI was diagnosed in 127 (25.1%). The areas under the curve of creatinine, CRP, and WBC to predict UTI were 0.41 (95% confidence interval [CI], 0.35-0.46), 0.71 (95% CI, 0.66-0.77), and 0.66 (95% CI, 0.60-0.72), respectively. The cutoffs were 0.26 mg/dL for creatinine, 2.3 mg/dL for CRP, and 14.4 × 103 cells/μL for WBC. Creatinine showed worse performance than the other variables. The application of creatinine added to the other variables led to an increase only in the sensitivity, but at the expense of a lower specificity, positive predictive value, and negative predictive value.
IntroductionFever is the most common chief complaint in children visiting emergency departments (EDs) [1]. Even after history taking and physical examination, 20% of such patients are classified as fever without source (FWS) [2]. With the introduction of heptavalent pneumococcal conjugate vaccine (PCV), the incidence of serious bacterial infection (SBI) among children with FWS has significantly declined [3].
Urinary tract infection (UTI) is the most common SBI among young children with FWS [4-6], and young children with UTI who develop pyelonephritis are put at the long-term sequelae of hypertension and renal failure [5,7,8]. Classic signs of UTI are usually not present in young children, instead fever being the most common symptom [9]. A different potential source of fever, such as otitis media, cannot rule out UTI [4,10]. Combined with the time-consuming task of obtaining clean urine specimens for culture, a reliable laboratory marker for predicting UTI would be of utmost value.
While many studies have reported white blood cell (WBC), absolute neutrophil count, and C-reactive protein (CRP) as surrogates for SBI [11,12], little has been verified for markers for UTI in the post-PCV era. A study has reported CRP to be superior to WBC in predicting confirmed bacterial infections, including Escherichia coli UTI [13]. CRP has also been reported to be useful in predicting bacteremic UTI in children [14-16].
An increase in serum creatinine concentration is a marker of acute kidney injury (AKI). AKI following severe UTI or dehydration may lead to the elevation of serum creatinine [17]. However, there have been no reports about the relationship between the UTI diagnosed at EDs and serum creatinine level. Several reports have proposed creatinine as a novel marker for predicting bacteremic UTI, especially in young infants [18,19]. Further validation, however, is yet to be conducted. Accuracy of creatinine in predicting UTI itself is also yet unclear. Thus, this study was conducted to assess the performances of serum creatinine, CRP, and WBC for predicting UTI in febrile children.
Methods1. Study design, setting, and populationThis was a retrospective analysis of a prospectively enrolled cohort of febrile children who presented to Seoul National University Hospital pediatric ED from August 2016 through February 2018. Our hospital is a large, urban, tertiary care, academic children’s hospital with an annual ED volume of more than 20,000 children encounters. This cohort registry (Fever Registry) included children aged 5 years or younger who visited the ED within 24 hours from the onset of fever (≥ 38.0。C). Among all children registered in the registry, children aged 24 months or younger were enrolled whose urine cultures were obtained. Those with missing values of creatinine, CRP or WBC were excluded. We also excluded children who had major comorbidities (e.g., congenital urinary tract malformations, immunodeficiency or congenital heart disease), who returned to the ED within 1 week from the prior visits, and who had SBIs other than UTI. The study protocol was approved by the Institutional Review Board (IRB no. 2001-155-1099), and written informed consent was waived due to a retrospective design.
2. Data collectionData from the registry included age and sex, comorbidities, temperature, heart rate, respiratory rate, history of antibiotics use in the 3 days preceding the visit, onset of fever, vaccination history, mental status, and severity of clinical manifestations. The manifestations included levels of feeding, activity, and urination on a degree of 0 (well) to 3 (poor), which were defined by the authors. A degree of clinical manifestation was determined by the caregivers’ answers to the questions about percentage of feeding, activity, and urination compared with normal state (0 = 75%-100%, 1 = 50%-75%, 2 = 25%-50%, 3 = 0%-25%). Laboratory findings included serum creatinine, CRP, and WBC, blood culture, urinalysis, urine culture, and cerebrospinal fluid profile and culture, and findings of chest radiograph. Hospitalized children were followed-up until discharge, and the final diagnoses were obtained.
3. OutcomesAccuracy of serum creatinine, CRP, and WBC in predicting UTI was the primary outcome. UTI was defined as the growth of a single urinary tract pathogen of at least 50,000 colony forming units/mL on a transurethral catheterized specimen.
4. Statistical analysisThe children were divided into the 2 groups according to diagnosis as UTI or non-SBI. Continuous variables were presented as medians with interquartile ranges, then compared by the Mann-Whitney U test. Categorical ones were compared by Fisher’s exact test. Receiver operating characteristic curves for predicting UTI were plotted for each laboratory marker. The optimal cutoffs for each marker were calculated based on the curves as the Youden method. For each marker’s optimal cutoffs, we reported sensitivity, specificity, positive and negative predictive values, and likelihood ratios with 95% confidence intervals (CIs). All statistical analyses were performed using STATA ver. 16.1 (StataCorp, College Station, TX). The level of statistical significance was defined as a P < 0.05.
Results1. Study populationDuring the study period, a total of 33,013 children visited the ED. Among the total visits, 7,847 (23.8%) febrile children aged 5 years or younger were registered to the Fever Registry. Of the 581 patients younger than 24 months whose urine culture was obtained, 506 met the inclusion criteria (Fig. 1). Among the 506 children, 127 (25.1%) had UTI, and the other 379 (74.9%) were classified as having non-SBI. Comparison of clinical characteristics between the 2 groups were summarized in Table 1. The children of the UTI group were significantly younger than the counterparts. Boys were more likely to have UTI. We found no significant difference with regards to temperature, heart rate, and respiratory rate. After the ED treatment, the children with UTI were more likely hospitalized than the counterparts.
2. Pathogens causing UTIPathogens isolated from the urine are listed in Table 2. E. coli was the most common pathogen (85.3%). All 5 children with bacteremic UTI had the bacterium as the pathogen.
3. Accuracy of creatinine, CRP, and WBCin identifying children with UTI The children of the UTI group had lower creatinine concentration, higher WBC counts and CRP concentration than those of the non-SBI group (Table 3). The receiver operating characteristic curves for creatinine, CRP, and WBC are shown in Fig. 2. The areas under the curve for creatinine, CRP, and WBC were 0.41 (95% CI, 0.35-0.46), 0.71 (95% CI, 0.66-0.77), and 0.66 (95% CI, 0.60-0.72), respectively. The optimal cutoffs were 0.26 mg/dL for creatinine, 2.3 mg/dL for CRP, and 14.4 × 103 cells/μL for WBC. The test characteristics of creatinine, CRP, and WBC at optimal cutoffs are presented in Table 4. CRP showed the highest overall predictive values with a positive and a negative likelihood ratios of 2.3 (95% CI, 1.9-2.7) and 0.4 (95% CI, 0.3-0.6), respectively. Creatinine showed poor performances than did WBC and CRP. The application of creatinine added to the other 2 tests led to an increase only in the sensitivity, but at the expense of a lower specificity, and positive and negative predictive values.
DiscussionIn this study, serum creatinine had the poorest accuracy in distinguishing UTI from non-SBI in the febrile children younger than 24 months. However, we found associations of higher levels of CRP and WBC with UTI in the children. While such changes are not definitive for final diagnosis, the elevations could be useful for predicting UTI. This is in line with a former study that reported increases in CRP and WBC levels in Finnish children with confirmed E. coli UTI [14]. Our finding confirms this association based on a population of children in the post-PCV era. In addition, we investigated novel cutoffs for CRP and WBC from the population.
To our best knowledge, the association between serum creatinine and UTI has not been previously reported. Several authors have found elevated creatinine concentration to be predictive of bacteremia in children with febrile UTI [18,19]. From our study population, creatinine showed a poor performance to predict UTI as well. However, we reported a possible contribution to sensitivity to the prediction. Significant elevations of serum creatinine are not apparent until 24-48 hours after the initiation of insult [17]. Therefore, the initial result of serum creatinine measured in EDs might be inappropriate to predict AKI induced by UTI despite the presence of relevant renal injury.
The low bacteremia rate in the children with UTI (3.9%) suggests a low feasibility of investigation on association between bacteremia and creatinine. Instead, we focused on the possible association of UTI with creatinine. Contrary to elevated creatinine concentration being predictive of bacteremic UTI [18,19], creatinine was not increased in the children with UTI than those with non-SBI. While creatinine concentration was minimally lower for those with UTI, this might be due to the younger median age of the UTI group [20]. The elevated creatinine in bacteremic UTI has been attributed to renal hypoperfusion secondary to a high infectious burden [18]. This was especially true for young infants in the first weeks of their life whose renal perfusion accounts for only 10% of the cardiac output [21]. This statement could not be validated by our study for the following reasons. First, the bacterial load for UTI patients was not high, as is shown by the low rate of bacteremia. Second, sub-analysis for very young infants was not conducted.
Our study had several limitations. First, it was a single-center study. As our hospital is a tertiary center located in the capital city of Korea, the features of the study population may not be readily generalizable. To enhance generalizability, we excluded all children with severe or rare underlying diseases affecting laboratory findings. Another limitation came from the retrospective design. Although the Fever Registry was a prospectively collected dataset, some parameters were unavailable, especially in the children with non-SBI. Lastly, our study included children younger than 24 months. For a more age group-specific analysis, a multi-center study may be needed to support our findings.
In conclusion, serum creatinine showed a poor performance to diagnose UTI in previously healthy febrile young children compared with CRP and WBC count. Since a single biomarker can neither rule in nor rule out UTI in febrile children, prediction of UTI can be achieved by interpretation of both clinical and laboratory findings.
Fig. 1.Flow diagram of the study population.ED: emergency department, SBI: serious bacterial infection, UTI: urinary tract infection.
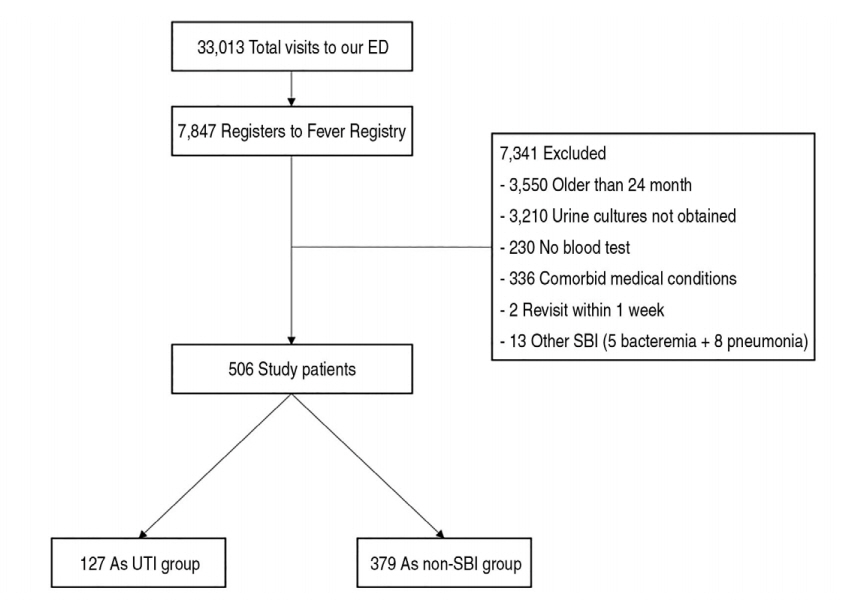
Fig. 2.AUCs for creatinine, CRP, and WBC to predict urinary tract infection. CRP: C-reactive protein, AUC: area under the curve, CI: confidence interval, WBC: white blood cell. 
Table 1.Clinical characteristics of the study population
Table 2.Pathogens in the children with UTI Table 3.Comparison of creatinine, CRP, and WBC between the children with and without UTI
Table 4.Test characteristics of creatinine, CRP, and WBC in predicting UTI
References1. Kwak YH, Kim DK, Jang HY. Utilization of emergency department by children in Korea. J Korean Med Sci 2012;27:1222–8.
2. Baraff LJ. Management of fever without source in infants and children. Ann Emerg Med 2000;36:602–14.
3. Greenhow TL, Hung YY, Herz A. Bacteremia in children 3 to 36 months old after introduction of conjugated pneumococcal vaccines. Pediatrics 2017;139:e20162098.
4. Hoberman A, Chao HP, Keller DM, Hickey R, Davis HW, Ellis D. Prevalence of urinary tract infection in febrile infants. J Pediatr 1993;123:17–23.
5. Chang SL, Shortliffe LD. Pediatric urinary tract infections. Pediatr Clin North Am 2006;53:379–400.
6. Shaw KN, Gorelick M, McGowan KL, Yakscoe NM, Schwartz JS. Prevalence of urinary tract infection in febrile young children in the emergency department. Pediatrics 1998;102:e16.
7. Benador D, Benador N, Slosman DO, Nussle D, Mermillod B, Girardin E. Cortical scintigraphy in the evaluation of renal parenchymal changes in children with pyelonephritis. J Pediatr 1994;124:17–20.
8. Winberg J. Commentary: progressive renal damage from infection with or without reflux. J Urol 1992;148:1733–4.
9. Ginsburg CM, McCracken GH Jr. Urinary tract infections in young infants. Pediatrics 1982;69:409–12.
10. Roberts KB, Charney E, Sweren RJ, Ahonkhai VI, Bergman DA, Coulter MP, et al. Urinary tract infection in infants with unexplained fever: a collaborative study. J Pediatr 1983;103:864–7.
11. Hsiao AL, Baker MD. Fever in the new millennium: a review of recent studies of markers of serious bacterial infection in febrile children. Curr Opin Pediatr 2005;17:56–61.
12. Bachur RG, Harper MB. Predictive model for serious bacterial infections among infants younger than 3 months of age. Pediatrics 2001;108:311–6.
13. Peltola V, Mertsola J, Ruuskanen O. Comparison of total white blood cell count and serum C-reactive protein levels in confirmed bacterial and viral infections. J Pediatr 2006;149:721–4.
14. Honkinen O, Jahnukainen T, Mertsola J, Eskola J, Ruuskanen O. Bacteremic urinary tract infection in children. Pediatr Infect Dis J 2000;19:630–4.
15. Gomez B, Mintegi S, Benito J, Egireun A, Garcia D, Astobiza E. Blood culture and bacteremia predictors in infants less than three months of age with fever without source. Pediatr Infect Dis J 2010;29:43–7.
16. Shaoul R, Lahad A, Tamir A, Lanir A, Srugo I. C reactive protein (CRP) as a predictor for true bacteremia in children. Med Sci Monit 2008;14:CR255–61.
17. Sutherland SM, Kwiatkowski DM. Acute kidney injury in children. Adv Chronic Kidney Dis 2017;24:380–7.
18. Averbuch D, Nir-Paz R, Tenenbaum A, Stepensky P, Brooks R, Koplewitz BZ, et al. Factors associated with bacteremia in young infants with urinary tract infection. Pediatr Infect Dis J 2014;33:571–5.
19. Megged O. Bacteremic vs nonbacteremic urinary tract infection in children. Am J Emerg Med 2017;35:36–8.
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||

 |
 |