 |
 |
AbstractDamage control resuscitation is a relatively new resuscitative strategy for patients with severe traumatic hemorrhage. This strategy consists of permissive hypotension and early balanced transfusion, and transfers the patients to subsequent surgery. There is growing evidence on harms of excessive fluids. Since 2013, survival benefit of massive transfusion protocol has been proven in adults. Despite insufficient evidence, pediatric massive transfusion protocols are widely used in North American trauma centers. This review focuses on the concept of damage control resuscitation, and summarizes the relevant pediatric evidence.
IntroductionDamage control resuscitation (DCR) is a resuscitative strategy for patients with severe traumatic hemorrhage to transfer them to subsequent surgery (Fig. 1). Recently, this strategy has been used as a standard therapeutic plan for severe traumatic hemorrhage in the U.S. and European countries [1-3]. Massive transfusion protocol (MTP) improves survival and hemostasis in adults. In children, efficacy of MTP remains a topic to debate. However, as of 2016, 92% of North American trauma centers were running a pediatric massive transfusion protocol (P-MTP) as a specific method of DCR [4]. This review focuses on the concept of DCR, and summarizes the relevant pediatric evidence.
Main subject1. Concept of DCRDCR, also known as hemostatic resuscitation, consists of permissive hypotension and early balanced transfusion. With these 2 key components, we can transfer patients with severe traumatic hemorrhage alive to subsequent surgery while minimizing the lethal triad (Fig. 1, 2). This concept suggests that such patients need relatively small volume of fluids and early transfusion with predefined amount and balance (Fig. 3). Of note, children have several anatomic and physiologic features to be considered when applying DCR (Table 1) [5].
The origin of DCR goes back to 1993 when Rotondo et al. [6] reported a survival benefit of the “damage control surgery.”This landmark study performed on 22 critically injured adults shows a higher survival rate in those who underwent damage control surgery compared to those who underwent definitive surgery (10 of the 13 adults vs. 1 of the 9 adults, P < 0.02). The surgical concept consists of 3 steps: (1) limited operations including control of bleeding and contamination, followed by packing and temporary closure of the abdomen (sometimes,“ open abdomen”); (2) resuscitation at intensive care units; and (3) definitive surgery [7,8].
2. Permissive hypotension1) ConceptPermissive hypotension, also known as hypotensive or controlled resuscitation, means a judicious fluid therapy to maintain systolic blood pressure slightly lower than age-adjusted low normal limits (70 + 2 × age in years mmHg). In adults, an arbitrary goal of systolic blood pressure may be 80-90 mmHg (in severe traumatic brain injury [TBI], 90-95 mmHg) [9]. Permissive hypotension is against a tenet of fluid therapy indicating the need for crystalloids 3 times the estimated blood loss (i.e., 3:1 rule) and 3 crystalloid boluses of 20 mL/kg (Fig. 3).
2) Theoretical harms of excessive fluidsExcessive fluids are potentially harmful to children in several ways [10-12]. First, increased hydrostatic pressure can dislodge clots, worsening hemorrhage. Second, dilution of coagulation factors and hypothermia can incur acute coagulopathy of trauma (ACOT) (Fig. 2). Third, cellular swelling leads to activation of inflammatory cytokines, resulting in end-organ injuries (e.g., abdominal compartment syndrome). Such harms had been supported mainly by animal studies [13].
3) Evidence for permissive hypotensionExcessive fluids should be avoided in children with severe hemorrhage. Recently, 3 adult systematic reviews show the lack of survival benefit of aggressive fluid therapy compared to permissive hypotension [14-16]. Harms of excessive fluids in children were suggested in a study performed on 907 children aged 14 years or younger who underwent transfusion at the U.S. military hospitals in Iraq and Afghanistan [16]. This study shows the association between large volume of crystalloids and increased length of stays at the intensive care units. In addition, large volume of crystalloids in the first 24 hours was associated with a higher mortality (18% [> 150 mL/kg] vs. 10% [≤ 150 mL/kg], P = 0.011) [17]. This evidence against excessive fluids is consistent with the deleted description of 3 crystalloid boluses in the tenth edition of the Advanced Trauma Life Support (Table 2) [18,19].
4) Caveats for pediatric applicationSevere TBI is the most common cause of death in childhood injury. In severe TBI, cerebral perfusion pressure (mean arterial pressure - intracranial pressure) should be kept at 50 mmHg or higher to minimize secondary brain injury [20]. Caution is needed when applying the concept of permissive hypotension to children with severe TBI. Penetrating torso injury (e.g., gunshot wounds), which is subject to excessive fluids-induced dislodgement of clots, can benefit more from judicious fluid therapy [21]. Thus, permissive hypotension should be used more rigorously to children with penetrating injury.
3. Early balanced transfusion1) ConceptEarly balanced transfusion means an empirical transfusion with sufficient amount and balance, mimicking whole blood. Besides TBI, injury can lead to death via hemorrhagic shock and ACOT that can be prevented with sufficient amount and balance of transfusion, respectively. This practice should be performed to children with severe traumatic hemorrhage, prior to recognition of the results of conventional coagulation tests (e.g., prothrombin time).
As a specific method of the early balanced transfusion, MTP denotes a pre-defined multidisciplinary transfusion protocol with the sufficient amount (any blood product ≥ 40 mL/kg/24 hour) and balance (ideally, packed red blood cell [PRBC]: fresh frozen plasma [FFP]:platelet [PLT] = 1:1:1) to provide an efficient DCR [12]. The definition of P-MTP is relatively arbitrary compared to MTP in adults (PRBCs ≥ 10 U/24 hour). A widely accepted recent definition of P-MTP is any blood product ≥ 40 mL/kg/24 hour that was driven from a study on children who visited the U.S. military hospitals in Iraq and Afghanistan [22]. Unlike permissive hypotension, children with severe TBI can benefit from MTP [23]. This feature may be due to the association between severe TBI and ACOT [24], and the need for large volume transfusion to minimize secondary brain injury [23].
2) Ideal whole blood and fatal ACOTChildren with severe traumatic hemorrhage may need MTP involving FFP infusion that mimics ideal whole blood to minimize fatal ACOT.
Whole blood has been used since World War I because it is readily available in combat settings, and includes all blood components required for oxygen delivery and hemostasis [25]. At war in Iraq and Afghanistan, warm and fresh whole blood transfusion shows survival benefit in adults with severe hemorrhage [26]. In children younger than 2 years, fresh whole blood may reduce post-open heart surgery hemorrhage [27]. However, such strengths of whole blood are offset by the short storage period of 21 days, leading to frequent use of fractionated blood products [25].
ACOT, also known as trauma-induced coagulopathy, is a collective term encompassing a wide range of systemic host defense dysregulation syndromes, manifesting various defects in hemostatic process [28]. This entity is aggravated by hypothermia, acidosis, dilution of coagulation factors, and fibrinolysis [29]. ACOT occurs in up to 25% at presentation, and increases mortality 4-fold [28]. A recent rat study using thromboelastography shows that induced hemorrhage was associated with significant increases in the time intervals to thrombin generation, without changes in the other time intervals to clot formation, fibrin cross-link, PLT aggregation, and fibrinolysis [30]. This rat study suggests the impaired thrombin generation as the main pathophysiology of ACOT, and may provide the rationale for FFP infusion in severe hemorrhage.
3) Evidence for P-MTPIn adults, MTP has proven efficacy for survival and hemostasis that were originally suggested by 2 randomized controlled trials [31,32]. Afterwards, many additional studies have supported the efficacy of MTP in adults [33-36]. However, it remains unknown on the activating trigger and ratio of blood products.
In contrast, we have only insufficient evidence on efficacy of P-MTP although many authors had tried to prove it (Table 3) [17,37-44]. The lack of proven survival benefit might be associated with low achievement rates of a 1:1 FFP:PRBC ratio (25%-37%) [17,39,43,44] and high TBI rates (30%-71%) [17,37,39-41,43,44] (Table 3). Also, it might stem from the inherent difficulties of study on critically injured children, such as the low incidence of events and ethical problems in obtaining informed consent. With the insufficient evidence, P-MTPs are performed largely based on extrapolation from adult studies as manifested by the higher median age of children receiving higher FFP:PRBC ratio (9-11 [≥ 1:2] vs. 4-7 years [< 1:2]) [42,43].
Recent literature about P-MTP has added evidence on increasing use of FFP, and its efficacy for hemostasis and survival (Table 3). Implementation of P-MTP is associated with a decrease in use of PRBC and an increase in use of FFP and PLT [40,42]. A single center study reported the absence of hemorrhagic deaths in 105 children undergoing P-MTP (mortality rate, 18.1%; all by TBI) [39]. A single center pre-post study on the implementation of P-MTP shows that all hemorrhagic deaths occurred in the pre-MTP group (mortality rate, 21.7%), indicating an improved hemostasis with MTP [40]. The same authors reported a survival benefit in 38 children receiving MTP with the relatively high median FFP:PRBC ratio of 0.9 [41]. In 2019, survival benefit of P-MTP was shown in the following 2 multicenter studies [43,44]. A study performed in 70 trauma centers (n = 465) shows that FFP:PRBC ratio ≥ 1:1 had a survival benefit (log rank, P = 0.02) [43]. According to the other study performed in 5 pediatric trauma centers (n = 110), the children receiving a 1:1 FFP:PRBC ratio had a lower mortality rate compared to those receiving a 1:2-3 FFP:PRBC ratio (15% vs. 29%-39%, P = 0.025) [44]. The latter study also shows that odds of mortality increased by 3.08 (95% confidence interval, 1.10-8.57) with each additional unit of PRBC deviation from a 1:1 FFP:PRBC ratio [44].
4) Caveats for pediatric applicationP-MTP is needed by only a portion of children with severe hemorrhage. Most injured children have relatively minor injury mechanisms (e.g., a minor fall) and more robust compensations against hypovolemic shock. Median frequency of activation was 6 times per year (interquartile range, 3-10) even in North American trauma centers running MTPs [4]. MTP has its own complications, such as hypothermia and coagulopathy [45]. In critically ill children, unnecessary PRBC transfusion can increase mortality [46]. Hence, controlled or minor hemorrhage is a contraindication for P-MTP.
5) Evidence summary and game planAs aforementioned, MTP in adults has a survival benefit without known trigger and ratio of blood products. Despite the insufficient evidence in children, in appropriate candidates, P-MTP should be performed considering its theoretical benefit, proven efficacy in adults, and recent pediatric evidence.
In the U.S. and Canada, P-MTPs are usually activated at the discretion of clinicians [4]. The triggers vary from 20-40 mL/kg/2 hour to 80 mL/kg/24 hour of anticipated volume of blood products [4]. In our opinion, a practical trigger and a target ratio of P-MTP may be an anticipated volume of blood products of 20-40 mL/kg in the first 2 hours and a 1:1-2 FFP:PRBC ratio, respectively. This speculation needs more evidence.
4. Application of DCR to the U.S. and Korean childrenTo investigate the use of DCR in reality, MTP was used as a surrogate for DCR given the more quantitative nature.
As of 2016, 92% of the U.S. and Canadian trauma centers are running P-MTPs [4]. Among these centers, 74% and 52% reported an FFP:PRBC ratio ≥ 1:2 and a PLT:PRBC ratio ≥ 1 (apheresis single-donor) or 5 (pooled random-donor):10, respectively. This survey suggests widespread use of P-MTP in North America with a wide variation in details about the use of blood products. This trend toward DCR in children is mentioned in the tenth edition of the Advanced Trauma Life Support (Table 2) [18,19].
In 2017, a Korean nationwide survey on MTP was performed at 48 hospitals of which the annual blood product use was 20,000 units or higher [47]. This survey shows that 15 hospitals (31.3%) had institutional MTPs (mostly, since 2012) without a mention of P-MTP, and of these, only 6 (12.5%) were actually running the protocols. In addition, P-MTP is not described in a manual for MTP published by the Korea Centers for Disease Control and Prevention [48]. Despite the recent suggestion of need for P-MTP [49], to our best knowledge, no such protocol has been established in Korea so far. Of note, Korean children need another caveat for pediatric application of DCR. Penetrating injury is less frequent in Korean children than in the U.S. children (2% [50] vs. 5% [51]), suggesting a smaller target of permissive hypotension in the former.
A Korean single center study on application of DCR to critically injured patients including children (age range, 13-87 years) shows that the patients with an FFP:PRBC ratio ≥ 1:2 had a higher 24-hour survival (72% vs. 97%, P < 0.001) compared to the counterpart without a difference in injury severity [52]. This study also shows that the former group received significantly less PRBC and crystalloids and more FFP and PLT in the first 24 hours.
ConclusionChildren with severe traumatic hemorrhage need DCR that is composed of judicious fluid therapy and early balanced transfusion with sufficient amount and balance to minimize the lethal triad. However, permissive hypotension should be applied cautiously to children with severe TBI. Survival benefit of MTP has been proven in adults. Despite the insufficient evidence in children, DCR, particularly P-MTP, should be performed in appropriate candidates considering its theoretical benefit, proven efficacy in adults, and recent pediatric evidence.
Fig. 1.A schematic diagram depicting the concept of DCR (hemostatic resuscitation) consisting of permissive hypotension and early balanced transfusion. It aims to transfer patients with severe traumatic hemorrhage to damage control surgery (or definitive surgery if appropriate) while bypassing or minimizing the lethal triad. As per this concept, such patients need judicious fluid therapy and early empirical transfusion with sufficient amount and balance. DCR: damage control resuscitation. 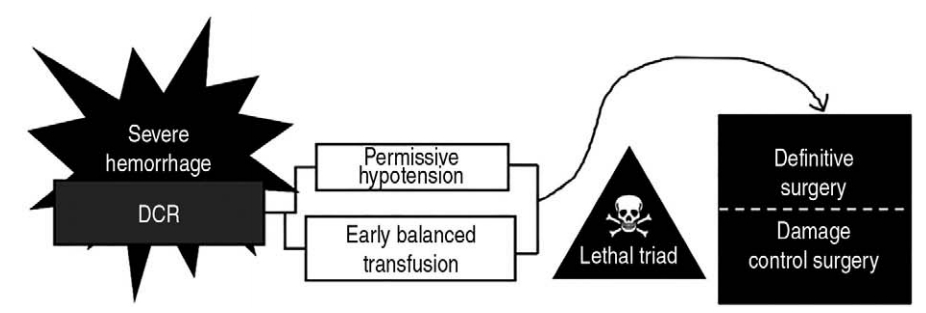
Fig. 2.The lethal triad of severe traumatic hemorrhage. This triangle acts as an obstacle to safe transfer to surgery by increasing mortality, particularly by ACOT. PRBC: packed red blood cell, ACOT: acute coagulopathy of trauma. 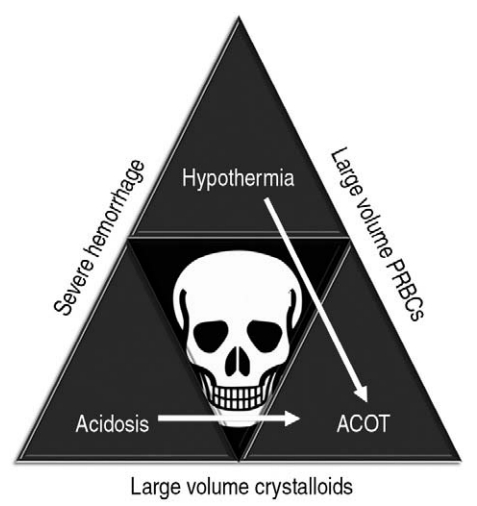
Fig. 3.DCR differs from the traditional 3 crystalloid boluses followed by PRBC infusion and/or additional transfusion. DCR: damage control resuscitation, PRBC: packed red blood cell, FFP: fresh frozen plasma, PLT: platelet, MTP: massive transfusion protocol. 
Table 1.Anatomic and physiologic features of children relevant to application of damage control resuscitation
Table 2.Table 3.
References1. Rossaint R, Bouillon B, Cerny V, Coats TJ, Duranteau J, Fernandez-Mondejar E, et al. The European guideline on management of major bleeding and coagulopathy following trauma: fourth edition. Crit Care 2016;20:100.
2. Cannon JW, Khan MA, Raja AS, Cohen MJ, Como JJ, Cotton BA, et al. Damage control resuscitation in patients with severe traumatic hemorrhage: a practice management guideline from the Eastern Association for the Surgery of Trauma. J Trauma Acute Care Surg 2017;82:605–17.
3. Galvagno SM Jr, Nahmias JT, Young DA. Advanced Trauma Life Support(®) update 2019: management and applications for adults and special populations. Anesthesiol Clin 2019;37:13–32.
4. Horst J, Leonard JC, Vogel A, Jacobs R, Spinella PC. A survey of US and Canadian hospitals’ paediatric massive transfusion protocol policies. Transfus Med 2016;26:49–56.
5. Nystrup KB, Stensballe J, Bottger M, Johansson PI, Ostrowski SR. Transfusion therapy in paediatric trauma patients: a review of the literature. Scand J Trauma Resusc Emerg Med 2015;23:21.
6. Rotondo MF, Schwab CW, McGonigal MD, Phillips GR 3rd, Fruchterman TM, Kauder DR, et al. ‘Damage control’: an approach for improved survival in exsanguinating penetrating abdominal injury. J Trauma 1993;35:375–82.
8. Tran A, Campbell BT. The art and science of pediatric damage control. Semin Pediatr Surg 2017;26:21–6.
9. Somand DM, Ward KR. Fluid and blood resuscitation in traumatic shock. Tintinalli JE, Stapczynski JS, Ma OJ, Yealy DM, Meckler GD, Cline DM, editors. Tintinalli’s emergency medicine: a comprehensive study guide 8th ed. New York (NY): McGraw-Hill Educations: 2016. p. 69–74.
10. Cotton BA, Guy JS, Morris JA Jr, Abumrad NN. The cellular, metabolic, and systemic consequences of aggressive fluid resuscitation strategies. Shock 2006;26:115–21.
12. Hughes NT, Burd RS, Teach SJ. Damage control resuscitation: permissive hypotension and massive transfusion protocols. Pediatr Emerg Care 2014;30:651–6.
13. Mapstone J, Roberts I, Evans P. Fluid resuscitation strategies: a systematic review of animal trials. J Trauma 2003;55:571–89.
14. Kwan I, Bunn F, Chinnock P, Roberts I. Timing and volume of fluid administration for patients with bleeding. Cochrane Database Syst Rev 2014;3:CD002245.
15. Albreiki M, Voegeli D. Permissive hypotensive resuscitation in adult patients with traumatic haemorrhagic shock: a systematic review. Eur J Trauma Emerg Surg 2018;44:191–202.
16. Tran A, Yates J, Lau A, Lampron J, Matar M. Permissive hypotension versus conventional resuscitation strategies in adult trauma patients with hemorrhagic shock: a systematic review and meta-analysis of randomized controlled trials. J Trauma Acute Care Surg 2018;84:802–8.
17. Edwards MJ, Lustik MB, Clark ME, Creamer KM, Tuggle D. The effects of balanced blood component resuscitation and crystalloid administration in pediatric trauma patients requiring transfusion in Afghanistan and Iraq 2002 to 2012. J Trauma Acute Care Surg 2015;78:330–5.
18. American College of Surgeons Committee on Trauma. Pediatric trauma. Merrick C, editor. Advanced trauma life support: student course manual 10th ed. Chicago (IL): American College of Surgeons: 2018. p. 186–212.
19. American College of Surgeons Committee on Trauma. Shock. Merrick C, editor. Advanced trauma life support: student course manual 10th ed. Chicago (IL): American College of Surgeons: 2018. p. 42–61.
20. American College of Surgeons Committee on Trauma. Head trauma. Merrick C, editor. Advanced trauma life support: student course manual 10th ed. Chicago (IL): American College of Surgeons: 2018. p. 102–27.
21. Bickell WH, Wall MJ Jr, Pepe PE, Martin RR, Ginger VF, Allen MK, et al. Immediate versus delayed fluid resuscitation for hypotensive patients with penetrating torso injuries. N Engl J Med 1994;331:1105–9.
22. Neff LP, Cannon JW, Morrison JJ, Edwards MJ, Spinella PC, Borgman MA. Clearly defining pediatric massive transfusion: cutting through the fog and friction with combat data. J Trauma Acute Care Surg 2015;78:22–8.
23. Rosenfeld EH, Lau P, Cunningham ME, Zhang W, Russell RT, Naik-Mathuria B, et al. Defining massive transfusion in civilian pediatric trauma with traumatic brain injury. J Surg Res 2019;236:44–50.
24. Talving P, Lustenberger T, Lam L, Inaba K, Mohseni S, Plurad D, et al. Coagulopathy after isolated severe traumatic brain injury in children. J Trauma 2011;71:1205–10.
25. Pidcoke HF, McFaul SJ, Ramasubramanian AK, Parida BK, Mora AG, Fedyk CG, et al. Primary hemostatic capacity of whole blood: a comprehensive analysis of pathogen reduction and refrigeration effects over time. Transfusion 2013;53:137S–49S.
26. Spinella PC, Perkins JG, Grathwohl KW, Beekley AC, Holcomb JB. Warm fresh whole blood is independently associated with improved survival for patients with combatrelated traumatic injuries. J Trauma 2009;66:S69–76.
27. Manno CS, Hedberg KW, Kim HC, Bunin GR, Nicolson S, Jobes D, et al. Comparison of the hemostatic effects of fresh whole blood, stored whole blood, and components after open heart surgery in children. Blood 1991;77:930–6.
28. Pohlman TH, Fecher AM, Arreola-Garcia C. Optimizing transfusion strategies in damage control resuscitation: current insights. J Blood Med 2018;9:117–33.
29. Hess JR, Brohi K, Dutton RP, Hauser CJ, Holcomb JB, Kluger Y, et al. The coagulopathy of trauma: a review of mechanisms. J Trauma 2008;65:748–54.
30. Harr JN, Moore EE, Wohlauer MV, Droz N, Fragoso M, Banerjee A, et al. The acute coagulopathy of trauma is due to impaired initial thrombin generation but not clot formation or clot strength. J Surg Res 2011;170:319–24.
31. Holcomb JB, del Junco DJ, Fox EE, Wade CE, Cohen MJ, Schreiber MA, et al. The prospective, observational, multicenter, major trauma transfusion (PROMMTT) study: comparative effectiveness of a time-varying treatment with competing risks. JAMA Surg 2013;148:127–36.
32. Holcomb JB, Tilley BC, Baraniuk S, Fox EE, Wade CE, Podbielski JM, et al. Transfusion of plasma, platelets, and red blood cells in a 1:1:1 vs a 1:1:2 ratio and mortality in patients with severe trauma: the PROPPR randomized clinical trial. JAMA 2015;313:471–82.
33. Hamidi M, Zeeshan M, Kulvatunyou N, Adun E, O’Keeffe T, Zakaria ER, et al. Outcomes after massive transfusion in trauma patients: variability among trauma centers. J Surg Res 2019;234:110–5.
34. Shrestha B, Holcomb JB, Camp EA, Del Junco DJ, Cotton BA, Albarado R, et al. Damage-control resuscitation increases successful nonoperative management rates and survival after severe blunt liver injury. J Trauma Acute Care Surg 2015;78:336–41.
35. Joseph B, Azim A, Zangbar B, Bauman Z, O’Keeffe T, Ibraheem K, et al. Improving mortality in trauma laparotomy through the evolution of damage control resuscitation: analysis of 1,030 consecutive trauma laparotomies. J Trauma Acute Care Surg 2017;82:328–33.
36. Hwang K, Kwon J, Cho J, Heo Y, Lee JC, Jung K. Implementation of trauma center and massive transfusion protocol improves outcomes for major trauma patients: a study at a single institution in Korea. World J Surg 2018;42:2067–75.
37. Hendrickson JE, Shaz BH, Pereira G, Parker PM, Jessup P, Atwell F, et al. Implementation of a pediatric trauma massive transfusion protocol: one institution’s experience. Transfusion 2012;52:1228–36.
38. Chidester SJ, Williams N, Wang W, Groner JI. A pediatric massive transfusion protocol. J Trauma Acute Care Surg 2012;73:1273–7.
39. Nosanov L, Inaba K, Okoye O, Resnick S, Upperman J, Shulman I, et al. The impact of blood product ratios in massively transfused pediatric trauma patients. Am J Surg 2013;206:655–60.
40. Hwu RS, Spinella PC, Keller MS, Baker D, Wallendorf M, Leonard JC. The effect of massive transfusion protocol implementation on pediatric trauma care. Transfusion 2016;56:2712–19.
41. Hwu RS, Keller MS, Spinella PC, Baker D, Tao Y, Leonard JC. Potential effects of high plasma to red blood cell ratio transfusion in pediatric trauma. Trauma 2017;19:21–7.
42. Cannon JW, Johnson MA, Caskey RC, Borgman MA, Neff LP. High ratio plasma resuscitation does not improve survival in pediatric trauma patients. J Trauma Acute Care Surg 2017;83:211–7.
43. Cunningham ME, Rosenfeld EH, Zhu H, Naik-Mathuria BJ, Russell RT, Vogel AM. A high ratio of plasma: RBC improves survival in massively transfused injured children. J Surg Res 2019;233:213–20.
44. Noland DK, Apelt N, Greenwell C, Tweed J, Notrica DM, Garcia NM, et al. Massive transfusion in pediatric trauma: an ATOMAC perspective. J Pediatr Surg 2019;54:345–9.
45. Lee JG. Massive transfusion in critically ill surgical patients. J Surg Crit Care 2012;2:14–7. Korean.
46. Kneyber MC, Hersi MI, Twisk JW, Markhorst DG, Plotz FB. Red blood cell transfusion in critically ill children is independently associated with increased mortality. Intensive Care Med 2007;33:1414–22.
47. Korea Centers for Disease Control and Prevention. The final report of the commission on evidence-based policymaking: development of standard operation manual for emergency and massive transfusion at hospitals [Internet]. Cheongju (Korea): Korea Centers for Disease Control and Prevention: c2018[cited 2019 Jun 17]. Available from: http://www.prism.go.kr/homepage/entire/retrieveEntireDetail.do?pageIndex=1&research_id=1351000-201700118&leftMenuLevel=160&cond_research_name=%EB%8C%80%EB%9F%89%EC%88%98%ED%98%88&cond_research_start_date=&cond_research_end_date=&pageUnit=10&cond_order=3. Korean.
48. Korea Centers for Disease Control and Prevention. Standard operation manual of massive transfusion [Internet]. Cheongju (Korea): Korea Centers for Disease Control and Prevention: c2018[cited 2019 Jun 17]. Available from: http://www.cdc.go.kr/CDC/together/CdcKrTogether0302.jsp?menuIds=HOME006-MNU2804-MNU3027-MNU2979&cid=138006. Korean.
49. Lee HT, Park PW, Seo YH, Ahn JY, Seo JY, Jeong JH, et al. Massive transfusion protocols for pediatric patients. Lab Med Online 2016;6:60–3. Korean.
50. Jung J, Eo E, Ahn K, Noh H, Cheon Y. Initial base deficit as predictors for mortality and transfusion requirement in the severe pediatric trauma except brain injury. Pediatr Emerg Care 2009;25:579–81.
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||

 |
 |