 |
 |
AbstractLaundry detergent pod (LDP) exposure has been reported to be fatal in children younger than 2 years, leading to respiratory or central nervous system depression. While gastrointestinal irritation is the most common symptom, there are reported cases of severe acidosis with respiratory depression or pneumonia, resulting in mortality. To our best knowledge, there is no report on a case of LDP exposure presenting with acute respiratory distress syndrome requiring extracorporeal membrane oxygenation support. Here, we present a case of a child with severe acute respiratory distress syndrome following LDP exposure, who was successfully treated with veno-pulmonary extracorporeal membrane oxygenation and steroids.
IntroductionLaundry detergent pod (LDP) exposure is most prevalent under 6 years of age, and can show various symptoms of gastrointestinal irritation, such as vomiting, and respiratory or central nervous system (CNS) depression (1,2). Compared to traditional detergents, LDP more frequently leads to severe toxicity (3,4). However, cases of life-threatening conditions have been rarely reported, such as acute respiratory distress syndrome (ARDS) with right heart dysfunction or severe acidosis. Herein, we present a pediatric case of ARDS following LDP exposure, which was successfully managed with lung-protective ventilation involving veno-pulmonary extracorporeal membrane oxygenation (ECMO) and steroid therapy. The patientвҖҷs legal guardian provided written consent for this case report that was approved by the international review board of Seoul National University Hospital (IRB no. H2305-028-1429).
CaseA 35-month-old boy presented to an outside emergency department (ED) with mental change and Kussmaul breathing. The boy was found with LDP all over his face and clothes by his father. Subsequently, he showed a sleeping tendency, and after 2 hours, he developed the abovementioned breathing pattern with cyanosis, prompting his mother to bring him to the ED. He was born at a gestational age of 36 weeks with a birth weight of 2.7 kg. After the birth, he was hospitalized at a neonatal intensive care unit for 3 weeks because of transient tachypnea. Otherwise, he had shown mild developmental delay suggesting an autism spectrum disorder.
At the outside ED, the initial vital signs were stable, including oxygen saturation. The initial arterial blood gas analysis, performed at ED about 30 minutes after his father recognized the exposure, showed the following results: pH, 7.086; PaCO2, 25.8 mmHg; PaO2, 100.5 mmHg; bicarbonate, 7.8 mmol/L; and lactate, 13.9 mmol/L. While waiting for detailed evaluation, the boy started to show bilateral mydriasis (5 mm/5 mm) and drooling with continuous cyanosis, leading to endotracheal intubation. Given the metabolic acidosis and an initial creatinine concentration of 0.68 mg/dL, a continuous renal replacement therapy was initiated and discontinued after correction of the acidosis later on day 2. Initial chest radiograph showed haziness on both lungs, which was dominant with patchy opacities on the right lung and later progressed to white-out (Fig. 1A, B). Methylprednisolone pulse therapy (30 mg/kg/day for 5 days) was performed against a rapid increase in the interstitial markings on the radiographs, indicating pneumonitis. He exhibited an abrupt drop in heart rate from 130-140 to 80 beats/minute with desaturation and underwent 1 cycle of cardiopulmonary resuscitation with 2 doses of epinephrine.
On day 2, C-reactive protein concentration was 4.7 mg/dL. Sedation was initiated to ensure effective mechanical ventilation. On day 3, bradycardia and desaturation recurred due to right pneumothorax. During the central venous catheter placement through the internal jugular vein, the aorta was presumed to be inadvertently punctured with the aortic valve injured (later turned out to have been inserted into the right proximal subclavian artery). On day 4, subcutaneous emphysema worsened, and pneumothorax with pneumomediastinum developed rapidly despite tube thoracostomy (Fig. 1C). As hypoxemia deteriorated and the boy rapidly developed ARDS with recurrent pneumothorax, veno-arterial ECMO was applied in the outside hospital (Fig. 1D). On day 8, he was transferred to our hospital and hospitalized directly to the intensive care unit for lung transplantation.
As the development of thrombi inside the ECMO circuit was uncontrollable even with maintenance of heparinization from the outside hospital, ECMO flow suddenly stopped, resulting in the repositioning of cannulas and changes in the circuit. A brain computed tomography showed lesions indicating global hypoperfusion injury rather than embolic lesions (Fig. 2A). Considering the edematous brain, we started to infuse mannitol, hypertonic saline, and dexamethasone as measures against increased intracranial pressure, and to monitor the pressure invasively. Incidentally, infarctions were noted in the right kidney and spleen (Fig. 2B-D).
Even after the cannula repositioning, circuit changes, and heparinization, thrombi formation persisted, and heparin was replaced with argatroban to rule out heparin-induced thrombocytopenia. No specific findings were noted in the evaluation of thrombophilia, including proteins C and S, anticardiolipin antibodies, lipoprotein (a), and homocysteine. Bronchoalveolar lavage was performed for evaluation and washing, which did not show evidence of infection, but CD8+ lymphocytes elevated up to 60% with a CD4+/CD8+ lymphocyte ratio of 35%/60%, consistent with hypersensitivity pneumonitis. Antiinflammatory doses of methylprednisolone were administered to the boy with hypersensitivity pneumonitis (Fig. 1E).
Veno-arterial ECMO was replaced with venopulmonary ECMO. This decision was based on the normal left ventricular function (ejection fraction, 60%) despite mild right ventricular dysfunction. If veno-venous ECMO had been used, hypoxemia would have been expected due to mixing with poorly oxygenated blood in the right atrium by severe impairment of the pulmonary function. To our best knowledge, this was the first pediatric application of veno-pulmonary ECMO in Korea. During management with lung-protective ventilation using nitric oxide and steroids, lung compliance and oxygenation rapidly improved in ARDS (Fig. 3). After 17 days from the transfer, ECMO was successfully weaned (Fig. 1F).
An electroencephalogram showed a normal sleep record, and the boy became alert with a Glasgow Coma Scale score of 13. Necrotic changes were found in previously infarcted lesions on follow-up brain magnetic resonance imaging. As the boy did not fully recover, he was transferred to the Department of Rehabilitation Medicine at the time of writing.
DiscussionLDP exposure can lead to variable clinical presentations and life-threatening conditions, such as lactic acidosis, respiratory depression, and CNS depression (5), especially in children younger than 2 years. They may exhibit drowsiness more frequently than older ones although frequently affected age is younger than 6 years (1,2,6,7). The pathogenesis of ARDS caused by LDP exposure may be associated with allergic responses, chemical pneumonitis, and surfactant effects, as suggested by reports in occupational medicine and toxicology (8-10). In cases of aspiration pneumonia with sepsis, 178 Pediatric antibiotics should be considered when infection cannot be ruled out to prevent mortality (7). In the present case, antibiotics were administered from the initial presentation at the outside hospital due to high concentrations of procalcitonin (136.4 ng/mL) and C-reactive protein (up to 11.4 mg/dL; data not shown). Banner et al. (7) reported on 2 pediatric patients who died of respiratory failure with severe acidosis and altered mental status, and suggested at least 4-hour monitoring for possible respiratory or CNS depression. In the present case, the pathophysiology of the ARDS remains unanswered. Aspiration may lead to chemical pneumonitis and can be a possible mechanism of ARDS. However, the case patient did not vomit despite the presence of LDP in his mouth, suggesting ingestion not leading to aspiration.
In ARDS, decreased fibrinolysis and increased coagulation activity can lead to a procoagulant state (11). Hypersensitivity pneumonitis may predispose patients to thromboembolism, possibly via inflammation and allergy-triggered activation of the coagulation cascade (12,13). In the present case, severe ARDS and suspicious hypersensitivity pneumonitis might have contributed to uncontrollable thrombosis at the transfer to our hospital.
A high index of suspicion for progression to ARDS and respiratory depression can guide early implementation of ECMO, resulting in an early, uncomplicated recovery. Among various types of ECMO, venopulmonary ECMO offers benefit in mixing with deoxygenated blood by delivering oxygenated blood directly to the pulmonary arteries, bypassing the right ventricle. Though the application of venopulmonary ECMO in ARDS is a rising option recently in adults, there is not an established consensus on the timing of application. Therefore, veno-venous ECMO is still a treatment of choice in severe ARDS. A complication requiring monitoring is pulmonary hemorrhage by increasing pulmonary arterial pressure. Further studies about indications and complications of veno-pulmonary ECMO are needed when we consider the incidence of right heart failure during the clinical course of severe ARDS in children (14,15).
Considering that respiratory or CNS depression can lead to mortality in children, it is crucial to keep LDP away from them and to prevent exposure, for example by banning the use of colorful and attractive packaging. The centralization of medical resources is an institutional problem that needs to be addressed when discussing critical care for patients from distant areas with ECMO. Further research is needed to provide statistics on the morbidity and mortality caused by respiratory failure, and based on the research, relevant guidelines for critical care should be developed.
NotesAuthor contributions Conceptualization: H Han, B Lee, and JD Park Methodology, Data curation, and Visualization: H Han Formal analysis and Investigation: WJ Jang, JW Lee, JH Kim, S Cho, and JD Park Supervision and Project administration: JD Park Writing-original draft: H Han Writing-review and editing: all authors All authors read and approved the final manuscript. Fig.В 1.Serial plain radiographs. (A) The initial radiograph shows a diffuse haziness and ground glass opacities, which are more remarkable on the right lung field, suggesting pneumonitis (day 1). (B) The right pulmonary lesion progressed to white-out (day 1). (C) Right pneumothorax (arrows) developed (day 3), and a chest tube (asterisk) was inserted (day 4). (D) Veno-arterial ECMO was implemented by draining the cannula into the inferior vena cava and returning the cannula into the ascending aorta (arrowheads) because of severe acute respiratory distress syndrome with recurrent pneumothorax (day 4). (E) After transfer to our hospital, steroid therapy was initiated for hypersensitivity pneumonitis, and cannulas for veno-pulmonary ECMO were inserted (arrowheads). (F) ECMO was successfully stopped 17 days after the transfer. ECMO: extracorporeal membrane oxygenation. 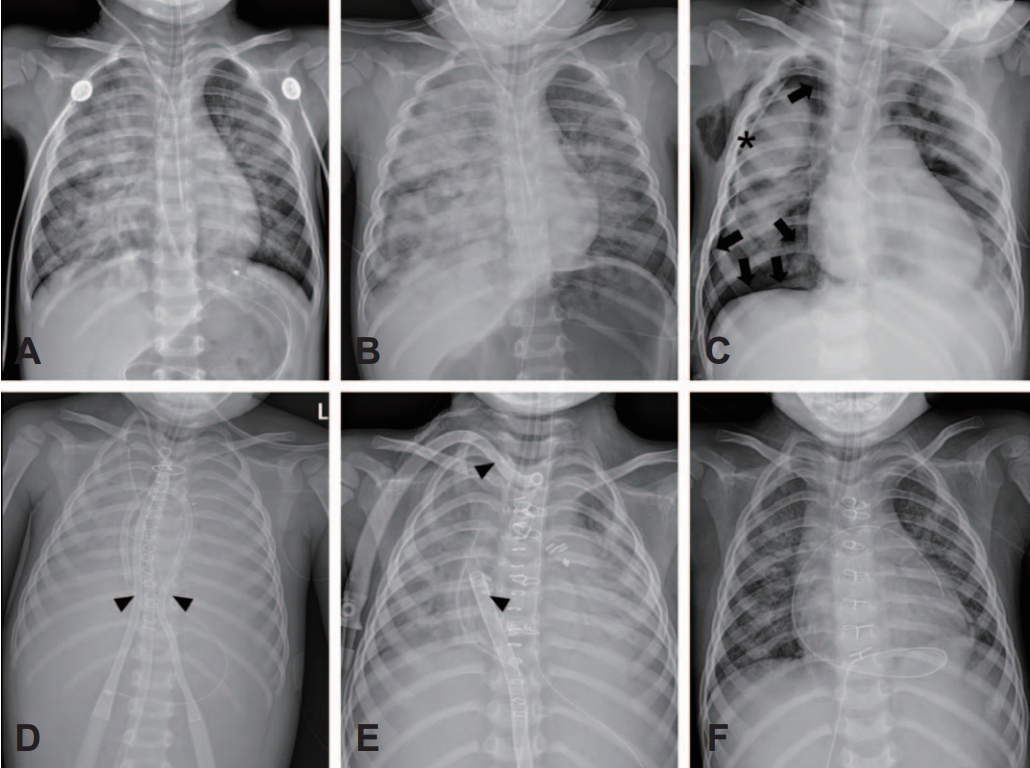
Fig.В 2.Multifocal infarcted lesions on computed tomography scans (after transfer). (A) Symmetrical wedge-shaped, hypodense lesions with internal hemorrhagic changes (asterisks) are found in bilateral watershed areas, with ventricles collapsed by the edematous brain. (B-D) Multifocal hypodense lesions are observed on the scans, which are consistent with probable infarctions in the right kidney and spleen (arrows). 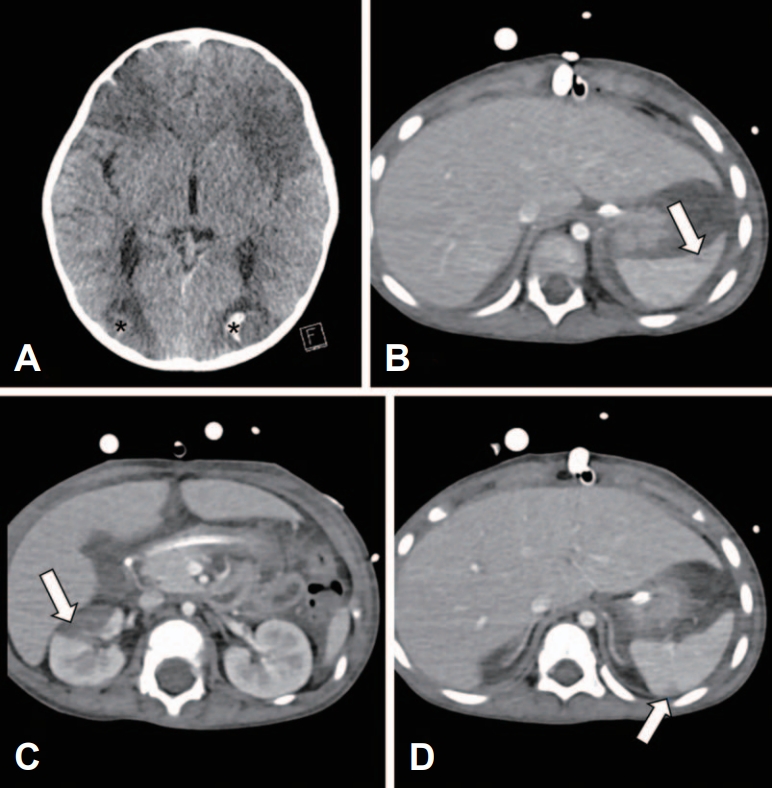
Fig.В 3.Trends of OI (line graph) and therapeutic interventions (bars with letters) for acute respiratory distress syndrome. Because OI (= [FiO2 Г— mean airway pressure] Г· PaO2) represents the degree of ventilator support needed to maintain oxygenation, change in OI after transfer to our hospital is plotted sequentially. OI: oxygenation index, NO: nitric oxide, VA ECMO: veno-arterial extracorporeal membrane oxygenation, ECMO: extracorporeal membrane oxygenation, HP: hypersensitivity pneumonitis. 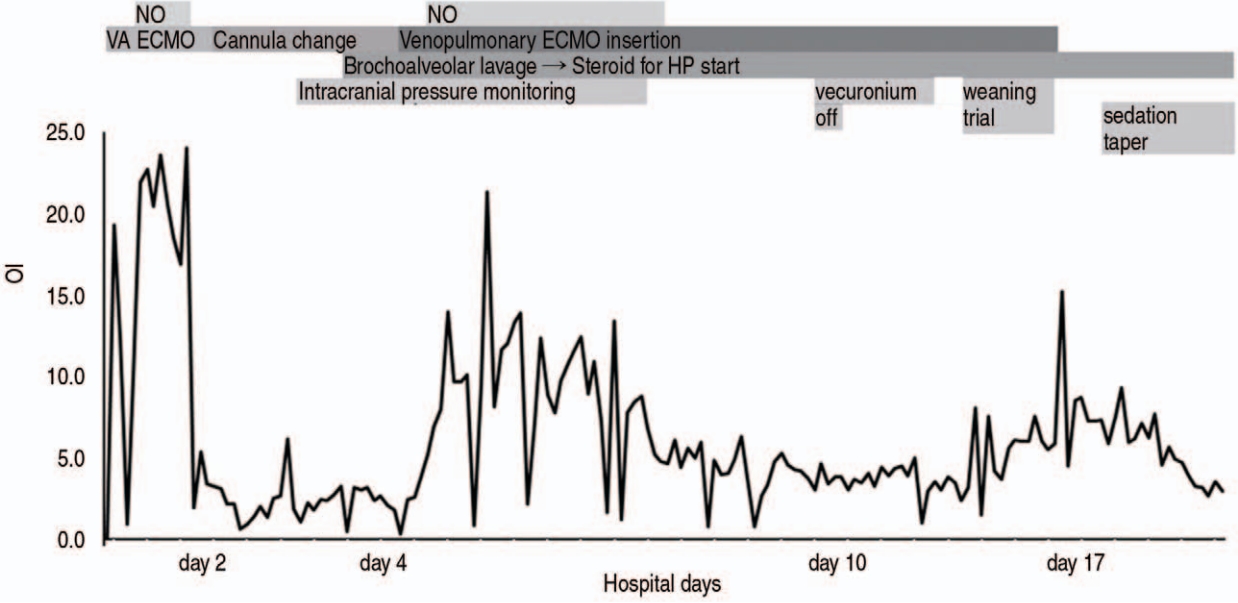
References1. Centers for Disease Control and Prevention (CDC). Health hazards associated with laundry detergent pods - United States, May-June 2012. MMWR Morb Mortal Wkly Rep 2012;61:825вҖ“9.
2. Roh DE. Laundry detergent pod: a rising cause of household poisoning. Pediatr Emerg Med J 2020;7:131вҖ“4.
3. Rigaux-Barry F, Patat AM, Cordier L, Manel J, Sinno-Tellier S. Risks related to pods exposure compared to traditional laundry detergent products: study of cases recorded by French PCC from 2005 to 2012. Toxicol Anal Clin 2017;29:257вҖ“66.
4. Settimi L, Giordano F, Lauria L, Celentano A, Sesana F, Davanzo F. Surveillance of paediatric exposures to liquid laundry detergent pods in Italy. Inj Prev 2018;24:5вҖ“11.
5. Day R, Bradberry SM, Thomas SHL, Vale JA. Liquid laundry detergent capsules (PODS): a review of their composition and mechanisms of toxicity, and of the circumstances, routes, features, and management of exposure. Clin Toxicol (Phila) 2019;57:1053вҖ“63.
6. Williams H, Bateman DN, Thomas SH, Thompson JP, Scott RA, Vale JA. Exposure to liquid detergent capsules: a study undertaken by the UK National Poisons Information Service. Clin Toxicol (Phila) 2012;50:776вҖ“80.
7. Banner W, Yin S, Burns MM, Lucas R, Reynolds KM, Green JL. Clinical characteristics of exposures to liquid laundry detergent packets. Hum Exp Toxicol 2020;39:95вҖ“110.
8. van Heemst RC, Sander I, Rooyackers J, de Jong L, Djamin RS, Aerts JG, et al. Hypersensitivity pneumonitis caused by occupational exposure to phytase. Eur Respir J 2009;33:1507вҖ“9.
9. Patel A, Hasany A, Tarlo SM. Occupational hypersensitivity pneumonitis after polyurethane adhesive exposure. CMAJ 2022;194:E1027вҖ“30.
10. Beuhler MC, Gala PK, Wolfe HA, Meaney PA, Henretig FM. Laundry detergentвҖң podвҖқingestions: a case series and discussion of recent literature. Pediatr Emerg Care 2013;29:743вҖ“7.
11. Nieuwenhuizen L, de Groot PG, Grutters JC, Biesma DH. A review of pulmonary coagulopathy in acute lung injury, acute respiratory distress syndrome and pneumonia. Eur J Haematol 2009;82:413вҖ“25.
12. Sobiecka M, Szturmowicz M, Lewandowska K, Kowalik A, ЕҒyzВҙwa E, Zimna K, et al. Chronic hypersensitivity pneumonitis is associated with an increased risk of venous thromboembolism: a retrospective cohort study. BMC Pulm Med 2021;21:416.
13. Lal A, Akhtar J, Pinto S, Grewal H, Martin K. Recurrent pulmonary embolism and hypersensitivity pneumonitis secondary to Aspergillus, in a compost plant worker: case report and review of literature. Lung 2018;196:553вҖ“60.
14. Zochios V, Yusuff H, Antonini MV, Schmidt M, Shekar K, for Protecting the Right Ventricle Network (PRORVnet). Veno-pulmonary arterial extracorporeal membrane oxygenation in severe acute respiratory distress syndrome: should we consider mechanical support of the pulmonary circulation from the outset? ASAIO J 2023;69:511вҖ“8.
|
|
|||||||||||||||||||||||||||||||||||

 |
 |