Continuous infusion of magnesium sulfate to treat acute severe asthma in a Brazilian pediatric emergency department
Article information
Abstract
Purpose
: Magnesium sulfate (MgSO4) emerged as an adjunctive therapy to help manage acute severe asthma refractory to the first-line therapy, such as oxygen, ipratropium, corticosteroids, and bronchodilators. Despite the known benefits of MgSO4, limited information is available regarding potential adverse effects (AEs). We aimed to investigate potential AEs and clinical response of intravenous MgSO4 by assessing vital signs and the modified Wood-Downes score (mWDS), a relevant clinical scoring system.
Methods
: This investigation constitutes a prospective, single-arm study, conducted from June 2022 through May 2023, in a pediatric emergency department of the quaternary public hospital in Brazil. This study included all children aged 3-14 years who visited the pediatric emergency department during the period and underwent a 6-hour continuous infusion of MgSO4 at 50 mg/kg/hour, against acute severe asthma, a case refractory to the first-line therapies. We evaluated the safety and efficacy of the infusion by using vital signs and the mWDS. Clinical response was defined as a reduction of at least 1 point in the scoring system.
Results
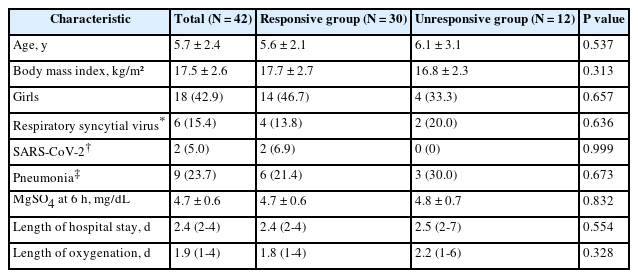
: We analyzed a total of 42 children including 24 boys (57.1%), whose mean age was 5.7 ± 2.4 years. The 6-hour continuous infusion of MgSO4 reduced mWDS from 3 (interquartile range, 3-4) to 2 (2-3) (P < 0.001) and resulted in the clinical response in 30 children (71.4%) without an AE. After the infusion, 13 of the 15 children (86.7%) classified as having moderate asthma exacerbation (mWDS, 4-6) were improved to having mild asthma exacerbation (mWDS, 1-3) (P < 0.001). The mean serum magnesium concentration after the infusion was 4.7 mg/dL. Pneumonia and viral infection did not predict the clinical response.
Conclusion
: This study indicates the safety and efficacy of the 6-hour continuous infusion of MgSO4 as a therapeutic option for acute severe asthma, which is refractory to the first-line therapy.
Introduction
The management of asthma in emergency departments (EDs) requires prompt action due to the potential reversibility of symptoms and the risk of respiratory failure. The standard treatment approach involves repeated administration of inhaled beta‐adrenergic agents, ipratropium, oxygen, and corticosteroids (1-4). In such instances, the use of intravenous magnesium sulfate (MgSO4) showed efficacy in improving asthma symptoms, but the assessment of its potential adverse effects (AEs) in children is limited (5-7).
MgSO4 induces smooth muscle relaxation leading to bronchodilation, and exhibits a rapid onset of action (4,5). To achieve a sustained spasmolytic effect, continuous infusion is required; otherwise, its use can be limited by its short half-life of 2.7 hours (8,9). The medication can be administered as a single bolus (10,11) or via continuous infusion for a more prolonged effect, as an attempt to compensate for its rapid renal elimination (2,12,13).
By investigating the AEs and evaluating clinical outcomes of continuous MgSO4 infusion, the authors aimed to examine whether continuous infusion for 6 hours for a more prolonged effect can be a therapeutic option in acute severe asthma in pediatric EDs. Findings from this study could help guide healthcare professionals in making informed decisions regarding the use of MgSO4 as an adjunctive therapy for severe acute asthma in children, and ultimately optimizing the emergency care and outcomes.
Methods
1. Study design and location
This prospective, single-arm study was carried out in a quaternary, public pediatric ED with 14 beds in Porto Alegre, Brazil, from June 2022 through May 2023. Prior to the administration of continuous infusion of MgSO4, legal guardians were provided with a document for review and approval. All legal guardians agreed to participate after reviewing the document. This study has been approved by the Research Ethics Committee of Hospital de Clínicas de Porto Alegre with a waiver for informed consent (IRB no. 23635618.1.0000.5327).
2. Selection of patients
The study included all children aged 3 to 14 years (upper age limit in the pediatric ED) who visited the pediatric ED during the study period and underwent a 6-hour continuous infusion of MgSO4 at 50 mg/kg/hour against acute severe asthma, which was defined as a case refractory to the first-line therapies. Exclusion criteria were a history of nephropathy, heart disease, chronic pulmonary diseases other than asthma, and genetic syndromes associated with pulmonary involvement.
During the infusion of MgSO4, all selected children were closely monitored and accompanied by emergency pediatricians. The prescription of MgSO4 was carried out by the attending physician in accordance with the institutional protocol. The 6-hour protocol was used as an attempt to increase the duration of the therapeutic effect and achieve better outcomes. The initial management included oxygen administration via a nasal cannula or Venturi mask based on saturation or respiratory distress, single-dose oral prednisolone, and rescue nebulization with salbutamol and ipratropium for 1-2 hours. Children who were refractory to the first-line therapy received the continuous infusion of 50 mg/kg/hour of MgSO4 for 6 hours (maximum, 8 g). None of them received MgSO4 prior to this intervention.
Vital signs were collected at 0, 2, 4, and 6 hours, and neurological signs were observed. Blood samples were drawn immediately after the completion of the 6-hour infusion to assess the serum magnesium concentrations. During the infusion of MgSO4, we closely monitored its AEs, such as nausea, drowsiness, vision changes, muscle weakness, hypotension, respiratory distress, arrhythmia, neurological disorders and burning sensation, or redness in the application route.
The reference for the vital signs, except blood pressure, was the American Heart Association (14). Hypotension was defined as systolic or diastolic blood pressure lower than the fifth percentile for age and sex. The children were classified by the modified Wood-Downes score (mWDS) (15,16). As described, the mWDS evaluates each child considering 5 clinical parameters: skin color, respiratory frequency, use of accessory musculature, pulmonary auscultation, and brain function. Each variable is classified from 0 to 2, varying the total score from 0 to 10 points. Scores 1-3 classify asthma attacks as mild, 4-6 moderate, and 7-10 severe (8). Clinical response was defined as a reduction of at least 1 point in the scoring system (16).
3. Demographic data and outcomes
Data were collected and recorded using the institutional REDCap system (Research Electronic Data Capture). The evaluated variables included age, sex, body mass index (BMI), serum magnesium concentration, presence of AEs, pneumonia, findings of viral testing for respiratory syncytial virus, severe acute respiratory syndrome coronavirus 2, adenovirus, influenza A (H1N1), and the clinical response measured by the mWDS (16). The outcomes were length of hospital stay, length of oxygenation, hospitalization to the pediatric intensive care unit (PICU), mechanical ventilation, and mortality.
4. Analysis
The collected data were analyzed using IBM SPSS Statistics ver. 20.0 (IBM Corp.). The Kolmogorov-Smirnov tests were used to assess the normality of variables. Categorical variables were presented as percentages and compared using the chi-square tests. Continuous variables with normal distribution were expressed as means with their respective standard deviations and compared using the Student’s t-tests. Continuous variables without asymmetrical distribution were expressed as medians with interquartile ranges and compared using the Mann-Whitney or Kruskal-Wallis tests. The Wilcoxon tests were used to assess changes over time. The McNemar tests were used to evaluate changes in before and after scores.
Results
The study included all children who were refractory to the first-line therapy in the ED, with no one meeting the exclusion criteria. The study population (n = 42) consisted of 24 boys (57.1%) and 18 girls (42.9%). Their mean age was 5.7 ± 2.4 years with a range of 3-12 years. The mean BMI showed the absence of overweight or obesity. After the 6-hour continuous infusion of MgSO4, the median value of mWDS decreased from 3 (interquartile range, 3-4) to 2 (2-3) (P < 0.001; Fig. 1).

Comparison of the modified Wood-Downes clinical score after the 6-hour continuous infusion of magnesium sulfate.
The clinical response after the infusion was observed in 30 children (71.4%) (Table 1). After the infusion, the conditions of 13 of the 15 children (86.7%) classified as moderate asthma attack (mWDS, 4-6) was improved to mild asthma attack (mWDS, 1-3) (P < 0.001). There was no difference between the responsive and unresponsive children, in terms of age, BMI, sex, virus testing, and pneumonia.
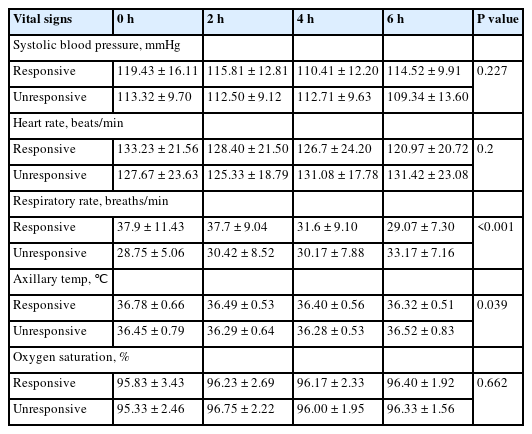
No AE was reported during or after the 6-hour continuous infusion. There were no differences between heart rate and systolic blood pressure during the infusion, but a significant reduction in respiratory rate was detected (Fig. 2, Appendix 1 [https://doi.org/10.22470/pemj.2024.00948]).

Charts of the variation in respiratory frequency during the infusion. Solid line indicates the responsive group, and dash line the unresponsive group.
The mean serum magnesium concentration after the infusion was 4.7 mg/dL. No differences were found between the preschoolers (aged 3-5 years) and schoolers (aged 6-12 years), in terms of the concentration and the percentage of response considering this stratification. The median length of hospital stay was 2.4 days (Table 1). It is noteworthy to mention that none of the children underwent hospitalization to the PICU or mortality.
Discussion
This study, which was performed on 42 children with acute severe asthma who underwent a 6-hour continuous infusion of MgSO4, showed a clinical response measured by an improved mWDS at the end of the infusion. The 6-hour infusion protocol showed no occurrence of AEs.
Since 1987, MgSO4 has been described as a safe medication to treat acute severe asthma, with a well-defined role of its infusion (12,13,17). A United States prospective cohort study using a 4-hour infusion showed improvement in tachycardia and tachypnea, and 3 of the 19 children who belonged to the intervention group reported mild AEs, such as nausea, vomiting, pain at the injection site, and flushing, not requiring infusion discontinuation or medical intervention (18). No differences between the intervention and control groups were observed in systolic or diastolic blood pressure and oxygen saturation (18). Another U.S. research, which was performed on 154 children hospitalized in a PICU and focused on the continuous infusion of MgSO4 for more than 24 hours, identified hypotension in 48.1% of the patients (17). The hypotensive events were mostly diastolic (94%) and limited to a single measurement of blood pressure (78%), and seldom required interventions (2.9%), such as fluids and reduction of the infusion. Continuous use for more than 24 hours may lead to the occurrence of AEs. However, in the present study, there was a reduction in the respiratory rate, probably due to clinical improvements.
The clinical response was observed in 71.4% of the study population, without identifying predictors of the unresponsiveness to the MgSO4 infusion. The unresponsiveness could be related to individual health conditions. Similar findings were observed by Irazuzta et al. (2,12) and Gross et al. (13), despite using an infusion for a shorter time (e.g., 4 hours). Recently, Forster et al. (19) analyzed a sample of 1,541 patients who received intravenous bolus of MgSO4 within 1 hour of the ED arrival, simultaneously to the first-line therapies, and no difference in hospitalization to the PICU was found. The use of bolus, instead of the continuous infusion, could be a factor of unresponsiveness due to the rapid renal clearance.
The AEs of MgSO4 are known as dose-dependent and are more commonly observed when serum magnesium concentrations exceed 9 mg/dL, higher than the 4.7 mg/dL observed in this present study (17). Although there is no consensus about the definition of toxic levels, Graff et al. (17) and Hardin et al. (20) observed that maintaining it between 4 and 6 mg/dL effectively prevented the occurrence of toxicity (9). This confirms that 4.7 mg/dL was far from the toxic level, and within therapeutic concentration, as previously mentioned. These findings indicate the safety and efficacy of the 6-hour continuous infusion of MgSO4 as a therapeutic option.
It is important to acknowledge that this research is performed in a single center, with a limited number of children. Despite this limitation, the results corroborate the findings of relevant research. Our findings indicate that the 6-hour continuous infusion of MgSO4 emerged as an interesting therapeutic option for the management of acute severe asthma, showing safe therapeutic serum concentrations without induction of toxicity while achieving clinical improvements. Further multicenter and randomized studies with larger sample sizes are recommended to enhance our understanding of its safety, efficacy, and AEs.
Notes
Author contributions
Conceptualization: DAD, PML, MGJ, JCBS, and LED
Data curation, Formal analysis, Software, Validation, and Visualization: DAD
Investigation: DAD, JVH, and JRSC
Methodology: DAD, PML, JCBS, and LED
Project administration: DAD, PML, and JCBS
Resources and Supervision: DAD and PML
Writing-original draft: DAD and PML
Writing-review and editing: DAD, PML, and JVH
All authors read and approved the final manuscript.
Conflicts of interest
No potential conflicts of interest relevant to this article were reported.
Funding sources
No funding source relevant to this article was reported.
Data availability
All data presented in this manuscript are available from the corresponding author upon reasonable request.